Thermodynamically controlled Diels–Alder reaction of 12-N-methylcytisine: A DFT study
Abstract
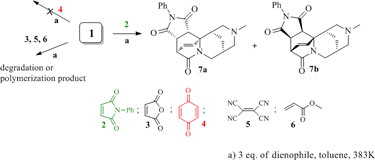
A DFT study was performed for the Diels–Alder traction of 12-N-methylcytisine with a number of dienophiles (in boiling toluene under atmospheric pressure), namely, N-phenylmaleimide, maleic anhydride, 2,4-benzoquinone, tetracyanoethylene and methyl methacrylate. It was shown that 12-N-methylcytisine selectively reacts with these dienophiles, only the reaction with N-phenylmaleimide (NPM) resulting in the formation of thermodynamically stable adducts, which is consistent with experimental data. This selectivity of 12-N-methylcytisine is attributable to the difference between the properties of the listed dienophiles, which is confirmed by the relative reactivity indices calculated within the framework of the frontier molecular orbital (FMO) and hard and soft (Lewis) acids and bases (HSAB) theories, the thermodynamic and activation parameters of the forward and retro-Diels–Alder reactions. According to analysis of the theoretical results, NPM is characterized by high chemical potential, hardness close to that of 12-N-methylcytisine, and commensurable heights of the activation barriers for the forward and reverse Diels–Alder reactions and also forms stable [4+2] adducts.